
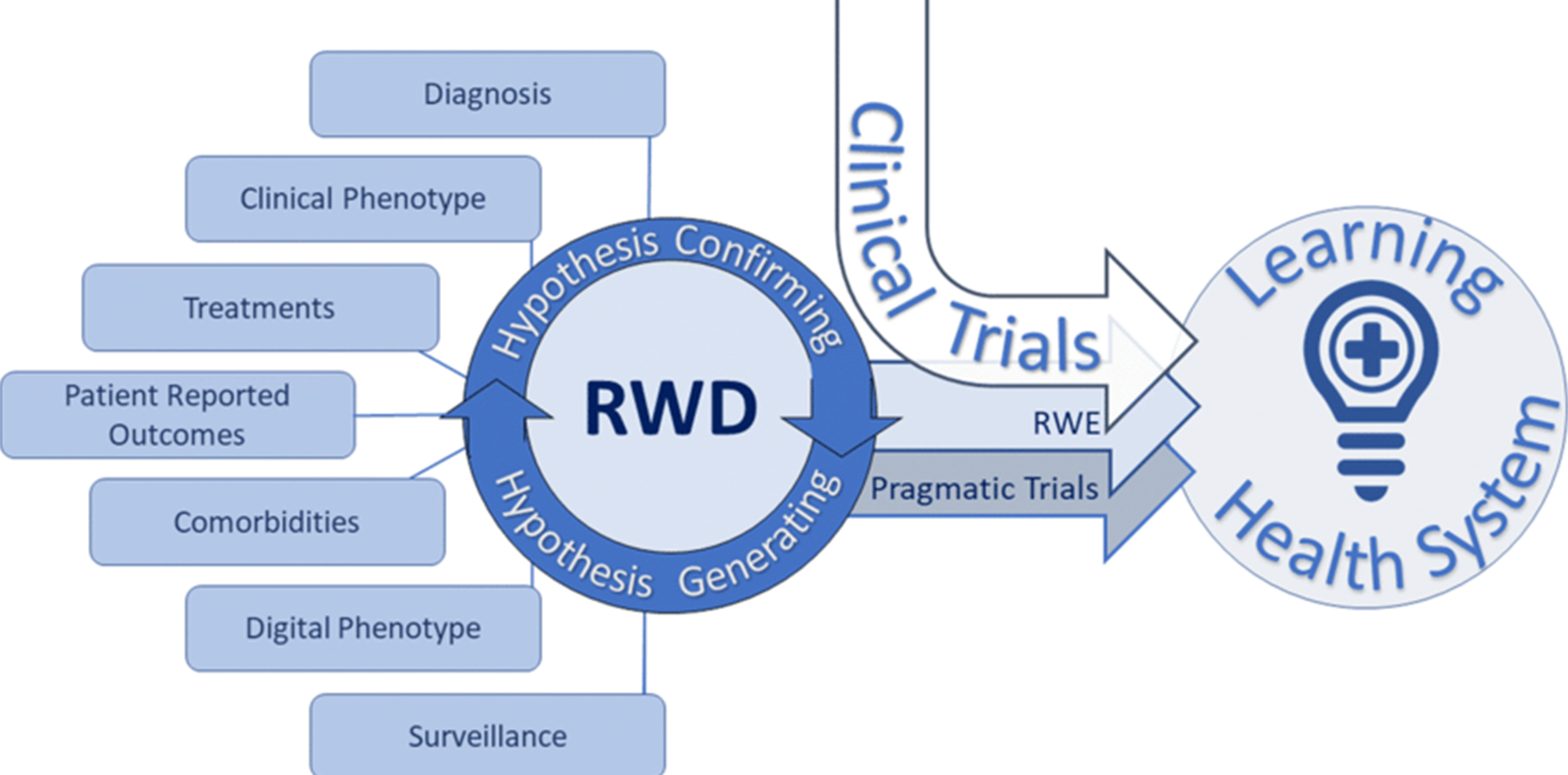
Real-Time Data and Real-World Evidence
At CLINLIA, we harness real-time data and real-world evidence (RWE) to enhance clinical decision-making and improve study outcomes. By integrating advanced analytics and real-world insights from everyday healthcare settings, we help sponsors evaluate treatment effectiveness, patient safety, and long-term outcomes — beyond the limitations of traditional clinical trials. Our approach brings data to life, driving faster, smarter, and more impactful research.
![]() Frequently Asked Questions:
Frequently Asked Questions:
1. What is Real-World Evidence and how does CLINLIA use it?
RWE refers to health data collected outside of traditional trials. We use it to support clinical insights, safety monitoring, and regulatory decisions.
2. Why is real-time data important in clinical research?
It enables faster decisions, early risk detection, and adaptive trial management — improving speed, accuracy, and outcomes.
